Video Lecture
Theory For Making Notes
Radioactivity
Becquerel unintentionally found in 1896 that uranyl potassium sulphate crystals release an invisible radiation that can discolour a photographic plate which was covered to block light. Following a series of tests, he came to the conclusion that the radiation the crystals were emitting was of a new kind and were so penetrating that they could affect the protected photographic plates and also could ionise the gases . Later on the term Radioactivity was used for these types of radiations which were produced due to spontaneous disintegration of heavy nuclei.
Other scientists’ investigations later revealed that some compounds have stronger radioactivity. The most important of these early studies was carried out by Marie and Pierre Curie. Following several years of meticulous and laborious chemical separation procedures on radioactive pitchblende ore, the Curies announced the discovery of two hitherto unidentified radioactive elements, polonium and radium. Further investigations, in the well-known alpha-particle scattering work by Rutherford, revealed that radioactivity results from the breakdown, or disintegration, of unstable nuclei.
Thus the phenomenon of spontaneous disintegration of heavy nuclei accompanied with the emission of \alpha , \beta{ }or{ } \gamma radiations is known as Radioactivity.
There are two types of radioactivity
Natural Radioactivity
This is exhibited naturally by certain heavy elements like uranium, radium, thorium, etc.
Induced or Artificial Radioactivity
Later it was found that the radioactivity can be induced in lighter elements using modern techniques. It is a process in which a stable nonradioactive nucleus is changed into a radioactive unstable nucleus by bombarding it with an appropriate particle like neutron, proton, alpha particle etc. Artificial radioactivity obeys the same laws as natural radioactivity.
Example : When Boron nuclei is bombarded with alpha particle of sufficient energy an isotope of nitrogen is formed which is radioactive with a half life of 9.9 minutes.
\displaystyle {}_{5}^{{10}}B+{}_{2}^{4}He\to {}_{7}^{{13}}N+{}_{0}^{1}n
\displaystyle {}_{7}^{{13}}N\to {}_{6}^{{13}}C+{}_{{+1}}^{0}\beta +\nu
Nuclear Stability
More than 1000 nuclides have been identified but not all are stable. If N be the number of neutrons and Z be the number of protons in a nuclei, then it is found that upto Z=20 those nuclei are stable in which N/Z ration is equal to 1 i.e. the number of protons and neutrons are equal. For heavy nuclides the ratio N/Z increases and becomes more than one i.e. in these nuclides the number of neutrons are more than the number of protons. All elements having Z > 83 are unstable and radioactive. The nuclides decay with time according to the laws of radioactive disintegration. They are called radioactive nuclide.
Radioactive decay- As written earlier, Stable nuclides have definite Z, N combinations. Nuclides with other Z, N combinations are also found in nature. However, these nuclides are unstable and they decay into other nuclides by various processes. Rutherford analyzed the radiations coming from radioactive sources and showed that these consists of three types of rays namely \displaystyle \alpha ,\beta \text{ and }\gamma rays.
So Three main processes by which an unstable nucleus decays are alpha decay, beta decay and gamma decay
Alpha Decay
The unstable nucleus \displaystyle _{z}^{A}X emits an alpha particle \displaystyle _{2}^{4}He in the alpha decay process and is converted into a daughter element \displaystyle _{{Z-2}}^{{A-4}}Y as given below.
\displaystyle _{z}^{A}X\to \text{ }_{{Z-2}}^{{A-4}}Y+\text{ }_{2}^{4}He
Note that in the \alpha decay the mass number of daughter element Y is 4 less than that of parent element X, where the atomic number is 2 less than that of parent element X
An example of alpha decay is
\displaystyle _{{83}}^{{212}}Bi\to _{{81}}^{{208}}{{T}_{1}}+_{2}^{4}He
The difference between the rest mass energy of the initial constituents and that of the final products is called the Q – value of the process
\displaystyle Q={{E}_{i}}-{{E}_{f}}
\displaystyle {{E}_{i}} is the rest mass energy of the initial constituents and \displaystyle {{E}_{f}} is that of the final products.
Beta decay
Beta decay is a process in which either a neutron is converted into a proton or a proton is converted into a neutron. Thus the ratio N/Z is altered in beta decay.
When a neutron is converted into a proton, an electron and a new particle named antineutrino are created and emitted from the nucleus.
\displaystyle n\to p+e+\bar{v}
Antineutrino is suppose to have zero rest mass, and charge less but has spin quantum number \displaystyle \pm {\scriptstyle{}^{1}\!\!\diagup\!\!{}_{2}\;}. The electron emitted from the nucleus is called a beta particle and is denoted by the symbol \displaystyle ({{\beta }^{-}}). The beta decay process may be represented by
\displaystyle _{Z}^{A}X\to _{{z+1}}^{A}Y+\bar{\beta }+\bar{v}\,
The rest mass energy of the initial constituents is
\displaystyle {{E}_{i}}=\left[ {m\left( {_{z}^{A}X} \right)-Z{{m}_{e}}} \right]{{c}^{2}}
and that of the final constituents is
\displaystyle {{E}_{f}}=\left[ {m\left( {_{{z+1}}^{A}Y} \right)-\left( {Z+1} \right){{m}_{e}}+{{m}_{e}}} \right]{{c}^{2}}
If the unstable nucleus has excess protons than for stability, a proton converts itself into a neutron. In the process, a positron and a neutrino are created and emitted from the nucleus
\displaystyle p\to n+{{e}^{+}}+v
This process is called Beta plus decay. The mass of a neutron is larger than the mass of a proton and hence the Q-value of such a process would be negative so, an isolated proton does not beta decay to a neutron.
Gamma Decay
When an alpha or a beta decay takes place, the daughter nucleus is generally formed in one of its excited states such a nucleus in an excited state eventually comes to ground state by emitting a photon or photons of electromagnetic radiation. The electromagnetic radiation emitted in nuclear transitions is called gamma ray.
In gamma decay neither the proton number nor the neutron number changes. Only the quantum states of the nucleons change.
Law of Radioactive Decay
The statistical radioactive law
In a typical radioactive decay an initially unstable nucleus called the parent, emits a particle and decays into a nucleus called the daughter, effectively, the birth of the daughter arises from the death of the parent. The daughter may be either the same nucleus in a lower energy state, as in the case of a \displaystyle \gamma -decay or an entirely new nucleus as arises from \displaystyle \alpha \text{ and }\beta decays. No matter what types of particles are emitted all nuclear decays follow the same radioactive decay law.
Alpha, beta and gamma decays are collectively called radioactive decay. If there are N active nuclei at an instant t. Then the rate of disintegration i.e. \displaystyle \frac{{dN}}{{dt}} will be proportional to N
\displaystyle \frac{{dN}}{{dt}}\propto N
\displaystyle \Rightarrow \frac{{dN}}{{dt}}=-\lambda N ( -ve sign indicates that the rate of disintegration \displaystyle \frac{{dN}}{{dt}} is decreasing with time)
\displaystyle \Rightarrow dN=-\lambda Ndt (The constant of proportionality \displaystyle \lambda is called decay constant)
\displaystyle \Rightarrow \frac{{dN}}{N}=-\lambda dt
\displaystyle \Rightarrow \int_{{{{N}_{0}}}}^{N}{{\frac{{dN}}{N}}}=-\lambda \int_{0}^{t}{{dt}}
hence \displaystyle N={{N}_{0}}{{e}^{{-\lambda t}}}
A plot of N Vs t of this equation is shown below
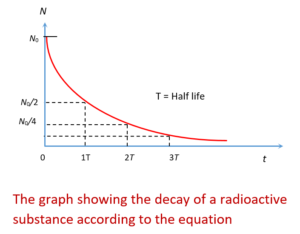
The equation is statistical, not a deterministic, law, it gives the expected number N of parent particles that survive after a time t. However for a large number of unstable nuclei, the actual number and expected number of survivors will almost certainly differ by no more than an insignificant fraction.
Graph of N Vs t on log scale
Consider N={{N}_{0}}{{e}^{{-\lambda t}}}
\Rightarrow \text{ }\frac{N}{{{{N}_{0}}}}={{e}^{{-\lambda t}}} taking natural log on both sides
we get \ln \frac{N}{{{{N}_{0}}}}= \ln({{e})^{-\lambda t}}
or \ln \frac{N}{{{{N}_{0}}}}=-\lambda t\ln (e)
or \ln \frac{N}{{{{N}_{0}}}}=-\lambda t (because ln(e)=1 )
or \ln (N) – \ln ({{N}_{o}}) = – \lambda t
hence \ln (N)= \ln ({{N}_{o}}) – \lambda t
Now the equation between ln(N) and t represent a straight line with negative slope as shown in the graph
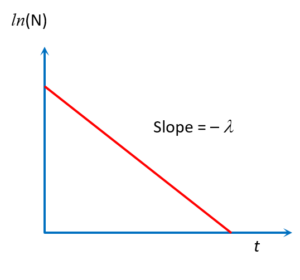
Note that the magnitude of inverse of slope (i.e. \frac{1}{\lambda } ) of \ln (N)\,\,vs\,\,t curve is known as mean life ( \displaystyle \tau ).
Activity
The quantity \displaystyle \left[ {-\frac{{dN}}{{dt}}} \right] gives the number of decays per unit time and is called the activity of the sample and denoted by A
Hence the equation \displaystyle -\frac{{dN}}{{dt}}=\lambda N can be written as A = \lambda N
If A(t) is activity at any time t then we can write
\displaystyle A(t)=\lambda N(t) Where N(t) is number of un-decayed particles at any time t
Similarly if {{A}_{o}} is the initial activity at t=0. Then \displaystyle A(0)=\lambda {{N}_{o}}, here {{N}_{o}} is the initial number of particles
Now dividing A(t) and {{A}_{o}} we get
\displaystyle \frac{{A(t)}}{{{{A}_{o}}}}=\frac{{N(t)}}{{{{N}_{o}}}}
or \displaystyle \frac{{A(t)}}{{{{A}_{o}}}}=\frac{{{{N}_{o}}{{e}^{{-\lambda t}}}}}{{{{N}_{o}}}}
hence \displaystyle A(t)={{A}_{0}}{{e}^{{-\lambda t}}}
SI unit of the activity is Becquerel which is the same as 1 disintegration/second
But popular unit of activity is curie
1 curie = \displaystyle 3.7\times {{10}^{{10}}} disintegration/s
Half-Life
The rapidity of decay of a particular radioactive sample is usually measured by the half-life {{T}_{1/2}}, defined as the time interval in which the number of parent nuclei at the beginning of the interval is reduced by a factor of one half.
Or It is the time required for the number of parent nuclei to fall to 50% of the initial number of nuclei i.e. \displaystyle \frac{{{{N}_{o}}}}{2}.
Calculation Of Half Life ( {{T}_{1/2}})
In the equation of decay i.e. \displaystyle N={{N}_{0}}{{e}^{{-\lambda t}}}
we put \displaystyle N=\frac{{{{N}_{o}}}}{2}
hence we get
\displaystyle \frac{{{{N}_{0}}}}{2}={{N}_{0}}{{e}^{{-\lambda {{t}_{{1/2}}}}}}
or \displaystyle \frac{1}{2}={{e}^{{-\lambda {{T}_{{1/2}}}}}}
taking natural log on both the sides we get
\displaystyle \ln (1)-\ln (2)=-\lambda {{T}_{{1/2}}}\ln (e)
so \displaystyle {{T}_{{1/2}}}=\frac{{\ln 2}}{\lambda }=\frac{{0.693}}{\lambda } ( because ln(1) = 0 , ln(e)=1 )
Important Points
1.
If N be the number of un-decayed particle after n half lives and {{N}_{o}} be the initial number of particles then N is given by \displaystyle N=\frac{{{{N}_{o}}}}{{{{2}^{n}}}}. Here \displaystyle n=\text{ number of half lives in the given time}=\frac{{Given\text{ time}}}{{\text{half life}}}
2.
Average Life
It is average of the lives of all the nuclei. It is defined as the sum of lives of all atoms divided by the total number of atoms
Hence \displaystyle {{T}_{{av}}}=\frac{{\text{Sum of the lives of all the atoms}}}{{\text{Total number of atoms}}}=\frac{{\int_{0}^{\infty }{{{{N}_{0}}{{e}^{{-\lambda t}}}dt}}}}{{{{N}_{0}}}}=\frac{1}{\lambda }=\frac{{{{T}_{{1/2}}}}}{{0.693}} = 1.44 {{T}_{1/2}}
Successive Disintegration and Radioactive Equilibrium
Suppose a radioactive element A disintegrates to form another radioactive element B which intern disintegrates to still another element C; such decays are called successive disintegration.
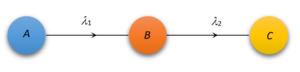
Rate of disintegration of A=\frac{{d{{N}_{1}}}}{{dt}}=-{{\lambda }_{1}}{{N}_{1}} (which is also the rate of formation of B)
Rate of disintegration of B=\frac{{d{{N}_{2}}}}{{dt}}=-{{\lambda }_{2}}{{N}_{2}}
Therefore net rate of formation of B = Rate of disintegration of A – Rate of disintegration of B
Hence net rate of formation of B = \displaystyle {{\lambda }_{1}}{{N}_{1}}-{{\lambda }_{2}}{{N}_{2}}
Equilibrium In Successive Disintegration
In radioactive equilibrium, the rate of decay of any radioactive product is just equal to it’s rate of production from the previous member.
i.e. \displaystyle {{\lambda }_{1}}{{N}_{1}}={{\lambda }_{2}}{{N}_{2}}
Hence \frac{{{{\lambda }_{1}}}}{{{{\lambda }_{2}}}}=\frac{{{{N}_{2}}}}{{{{N}_{2}}}}=\frac{{{{{({{T}_{{1/2}}})}}_{2}}}}{{{{{({{T}_{{1/2}}})}}_{1}}}}
Illustration
The half-life of Cobalt – 60 is 5.25 years. How long after its activity have decreased to about one-eigth of its original value ?
Solution
The activity is proportional to the number of undecayed atoms.
In each half-life, half the remaining sample decays.
Since \displaystyle \left( {\frac{1}{2}} \right) \times \left( {\frac{1}{2}} \right) \times \left( {\frac{1}{2}} \right)={{\left(\frac{1}{8}\right)}^{th}},
therefore, three half-lives or 15.75 years are required for the sample to decay to \displaystyle \frac{1}{8}th its original strength.
Illustration
A count rate-meter is used to measure the activity of a given sample. At one instant the meter shows 4750 counts per minute. Five minutes later it shows 2700 counts per minute.
(a) Find the decay constant
(b) Also, find the half life of the sample
Solution
Initial Activity
{{A}_{i}} = {{\left. {\frac{{dN}}{{dt}}} \right|}_{{t=0}}}=\lambda {{N}_{o}}=4750 …(i)
Final Activity
{{A}_{f}}= {{\left. {\frac{{dN}}{{dt}}} \right|}_{{t=5}}}=\lambda N=2700\,\, …(ii)
Dividing (i) by (ii), we get
\frac{{4750}}{{2700}}=\frac{{{{N}_{o}}}}{{{{N}_{{t\,}}}}}
The decay constant is given by
\displaystyle \lambda = \frac{{2.303}}{t}\log \frac{{{{N}_{o}}}}{{{{N}_{t}}}}
or \displaystyle \lambda = \frac{{2.303}}{5}\log \left(\frac{{4750}}{{2700}}\right)
on solving we get \lambda = 0.113{ }{{\min }^{-1}}
Hence half life of the sample is
T = \frac{{0.693}}{\lambda }=\frac{{0.693}}{{0.113}}
Hence T = 6.14 min
Illustration
The mean lives of a radio active substance are 1600 and 400 years for \displaystyle \alpha – emission and \displaystyle \beta – emission respectively. Find out the time during which three fourth of a sample will decay if it is decaying both by \displaystyle \alpha – emission and \displaystyle \beta – emission simultaneously.
Solution
When a substance decays by \displaystyle \alpha and \displaystyle \beta emission simultaneously, the average disintegration constant \displaystyle {{\lambda }_{{av}}} is given by
\displaystyle {{\lambda }_{{av}}}={{\lambda }_{\alpha }}+{{\lambda }_{\beta }}
where \displaystyle {{\lambda }_{\alpha }}= disintegration constant for \displaystyle \alpha – emission only
and \displaystyle {{\lambda }_{\beta }}= disintegration constant for \displaystyle \beta – emission only
Mean life is given by \displaystyle {{T}_{m}}=\text{ }1/\lambda
Hence \displaystyle {{\lambda }_{{av}}}={{\lambda }_{\alpha }}+{{\lambda }_{\beta }}
or \frac{1}{{{{T}_{m}}}}=\frac{1}{{{{T}_{\alpha }}}}+\frac{1}{{{{T}_{\beta }}}}
so \frac{1}{{T}_{m}}= \frac{1}{{1600}}+\frac{1}{{400}} = 3.12 \times {{10}^{-3}}
{{\lambda}_{av}}t = 2.303 log \frac{{{{N}_{o}}}}{{{{N}_{t}}}}
hence (3.12 \times {{10}^{-3}})t = 2.303 log \frac{{100}}{{25}}
so t = 2.303 \times \frac{1}{{3.12\,\times {{{10}}^{{-3}}}}}\log 4 = 443.5 years
Illustration
The half-life of radium is 1620 years. How many radium atoms decay in 1s in a 1g sample of radium. The atomic weight of radium is 226 kg/mol. (Take one year = 3.16 \times {{10}^{7}} s )
Solution
Number of atoms in 1 g sample is
\displaystyle N = \left( {\frac{{0.001}}{{226}}} \right)\left( {6.02\times {{{10}}^{{26}}}} \right)
hence N = 2.66\times {{10}^{{21}}} atoms.
The decay constant is \lambda = \frac{{0.693}}{{T}_{1/2}}
therefore \lambda =\frac{{0.693}}{{\left( {1620} \right)\left( {3.16\times {{{10}}^{7}}} \right)}}
hence \lambda=1.35\times {{10}^{{-11}}}{{s}^{-1}}
Since, \frac{{dN}}{{dt}}=\lambda N
therefore \frac{{dN}}{{dt}} =\left( {1.35\times {{{10}}^{{-11}}}} \right)\left( {2.66\times {{{10}}^{{21}}}} \right)
hence \frac{{dN}}{{dt}} = 3.6\times {{10}^{{10}}}{{s}^{-1}}
Thus, 3.6 \times {{10}^{10}} nuclei decay in one second.
Illustration
What is the activity of one gram of _{{88}}^{{226}}Ra\,, whose half-life is 1622 years?
Solution
The number of atoms in 1g of radium is
N=(1\,\text{g})\ \left( {\frac{{1\,\text{g}-\text{mole}}}{{\text{226}\ \text{g}}}} \right)\ \left( {6.025\times {{{10}}^{{23}}}\ \frac{{\text{atoms}}}{{\text{g}-\text{mole}}}} \right)
hence N=2.666\times {{10}^{{21}}}
The decay constant is related to the half-life by the relation \lambda =\frac{{0.693}}{{{{T}_{{1/2}}}}}
hence \displaystyle \lambda =\frac{{0.693}}{{1622}}yea{{r}^{{-1}}}
or \displaystyle \lambda =\frac{{0.693}}{{1622\times 365\times 24\times 3600}}{{\sec }^{{-1}}}
or \displaystyle \lambda =1.355\times {{10}^{{-11}}}{{\mathbf{s}}^{{-1}}}
The activity is then found from
Activity = \displaystyle \lambda N=\text{ }(1.355\times {{10}^{{-11}}}{{s}^{{-1}}})\text{ }(2.666\times {{10}^{{21}}})
hence Activity = \displaystyle 3.612\times {{10}^{{10}}} disintegration/s
The definition of the curie is 1Curie = \displaystyle 3.7\times {{10}^{{10}}} disintegrations/s. This is approximately equal to the value found above.
Illustration
There are two radioactive nuclei A and B. A is an alpha emitter and B is a beta emitter. If their disintegration constants are in the ratio 1 : 2, then the ratio of number of atoms of A and B at any time t so that probabilities of getting alpha and beta particles are same at that instant must be
(a) 2 : 1
(b) 1 : 2
(c) e
(d) {{e}^{-1}}
Solution
\frac{{{{\lambda }_{A}}}}{{{{\lambda }_{B}}}}=\frac{1}{2}
Probabilities of getting \displaystyle \alpha \text{ and }\beta particles are same. Thus rate of disintegration are equal
hence \displaystyle {{\lambda }_{A}}{{N}_{A}}={{\lambda }_{B}}{{N}_{B}}
or \frac{{{{N}_{A}}}}{{{{N}_{B}}}}=\frac{{{{\lambda }_{B}}}}{{{{\lambda }_{A}}}}=2
so option (a) is correct
Illustration
A radioactive material has half life’s for \alpha and \beta emission equal to 20 and 100 yrs respectively. \frac{1}{8}th fraction of the radioactive material will be remain there after
(a) 360 yrs
(b) 50 yrs
(c) 120 yrs
(d) 180 yrs
Solution
As we know for a sample \lambda ={{\lambda }_{1}}+{{\lambda }_{2}}
or \displaystyle \frac{1}{T}=\frac{1}{{{{T}_{1}}}}+\frac{1}{{{{T}_{2}}}} (because \lambda = \frac{1}{T})
hence T=\frac{{{{T}_{1}}{{T}_{2}}}}{{{{T}_{1}}+{{T}_{2}}}}
or T=\frac{{20\times 100}}{{20+100}}=\frac{{200}}{{12}}=\frac{{50}}{3}yrs
also, \frac{N}{{{{N}_{0}}}}={{\left( {\frac{1}{2}} \right)}^{{\frac{t}{T}}}}
{ } \Rightarrow \frac{1}{8}={{\left( {\frac{1}{2}} \right)}^{{\frac{t}{T}}}}
or \displaystyle {{\left( {\frac{1}{2}} \right)}^{3}}={{\left( {\frac{1}{2}} \right)}^{{\frac{t}{T}}}}
therefore \frac{t}{T}=3\Rightarrow t=3T=\frac{{50\times 3}}{3}=50yrs
so the correct option is (b)
Illustration
The probability of a radioactive atom for not disintegrating till 3 times of its half life is
(a) 1/3
(b) 1/4
(c) 1/8
(d) 7/8
Solution
Probability = (not disintegrate for \displaystyle {{1}^{{st}}} half) × (not disintegrate in \displaystyle {{2}^{{nd}}} half) × (not disintegrated in \displaystyle {{3}^{{rd}}} half
so Probability = \displaystyle {\scriptstyle{}^{1}\!\!\diagup\!\!{}_{2}\;}\times {\scriptstyle{}^{1}\!\!\diagup\!\!{}_{2}\;}\times {\scriptstyle{}^{1}\!\!\diagup\!\!{}_{2}\;}= \frac{1}{8}
hence option (c) is correct
Practice Questions (Basic Level)
1.
The energy of neutron in thermal equilibrium at room temperature is approximately
(a) 5 eV
(b) 1 eV
(c) 0.1 eV
(d) 0.025 eV.
Ans (d)
2.
1 milligram radium has 2.68 × 1018 nuclei. Its half-life is 1620 year. After 3240 year, how many nuclei would have disintegrated?
(a) 1.82 × 1018
(b) 1.34 × 1018
(c) 0.67 × 1018
(d) 2.01 × 1018.
Ans (d)
3.
Fast neutrons can easily be slowed down by
(a) the use of lead shielding
(b) passing them through heavy water
(c) elastic collision with heavy nuclei
(d) applying strong magnetic field.
Ans (b)
4.
What percentage of a radioactive substance is left after 5 half-live
(a) 31%
(b) 3.12%
(c) 0.3%
(d) 1%
Ans (b)
5.
Radon has 3.8 day as its half-life. How much radon will be left out of 15 mg mass after 38 day?
(a) 1.515 mg
(b) 0.015 mg
(c) 1.049 mg
(d) 9.327 mg
Ans (b)
Practice Questions (JEE Main Level)
1.
An element A decays into element C in two steps as
\text{A}\to \text{B}+{{\,}_{\text{2}}}\text{H}{{\text{e}}^{\text{4}}}and \text{B}\to \text{C}+{{2}_{{-\text{1}}}}e{}^\circ then,
(a) A and C are isotopes
(b) A and C are isobars
(c) A and B are isotopes
(d) A and B are isobars.
Ans (a)
2.
A stationary radioactive nucleus of mass 210 unit disintegrates into an alpha particle of mass 4 unit and residual nucleus of mass 206 unit. If the kinetic energy of the alpha particle is E, the kinetic energy of the residual nucleus is
(a) \left( {\frac{2}{{105}}} \right)\,E
(b) \left( {\frac{2}{{103}}} \right)\,E
(c) \left( {\frac{{103}}{{105}}} \right)\,E
(d) \left( {\frac{{103}}{2}} \right)\,E.
Ans (b)
3.
At any instant, the ratio of amount of radioactive substances is 2 : 1. If their half-lives be respectively 12 and 16 hours, then after two days, what will be the ratio of substances?
(a) 1 : 1
(b) 2 : 1
(c) 1 : 2
(d) 1 : 4
Ans (d)
4.
After a certain lapse of time, fraction of radioactive polonium undecayed is found to be 12.5% of the initial quantity. What is the duration of this time lapsed if the half life of polonium is 138 days?
(a) 124 day
(b) 414 day
(c) 345 day
(d) 534 day
Ans (b)
5.
For every 106 atoms of radium in a sample today, find the number of atoms that will be left after 3200 year. Assume half-life of radium to be 1600 year.
(a) 0.5 × 105 atoms
(b) 3.7 × 105 atoms
(c) 2.5 × 105 atoms
(d) 1.3 × 105 atoms
Ans (c)
6.
The radioactive isotope _{{27}}^{{60}}Co is used in the treatment of tumors. It undergoes
Β decay with a half-life of 5.25 y. What is the initial decay rate of a 0.01 g sample?
(a) 0.2 x 1011Bq
(b) 3.7 x 1011Bq
(c) 4.2 x 1011Bq
(d) 5.7 x 1011Bq
Ans (c)
7.
The decay rate of a freshly prepared sample is 15 \displaystyle \mu Ci. It drops to 9 \displaystyle \mu Ci after 2.5 h.
(a)
Find the half-life of the nuclide.
(a) 3.9 h
(b) 5.7 h
(c) 2.8 h
(d) 3.4 h
Ans (d)
(b)
How many radioactive atoms were initially present?
(a) 2.7 x 109
(b) 9.8 x 109
(c) 0.3 x 109
(d) 1.5 x 109
Ans (b)
Practice Questions (JEE Advance Level)
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
